Microbial diversity in the surroundings and selected parts of the imperial throne of D. Pedro II - characterization by classical microbiology and molecular biology, before and after restoration interventions
DOI:
https://doi.org/10.14568/cp2019020Keywords:
D. Pedro’s II Throne, Bacteria, Fungi, Molecular Biology, Microbial DiversityAbstract
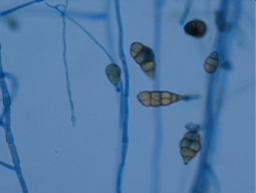
The Museu Imperial has an important historical collection from the Portuguese Royal Family in Brazil. The throne represents the imperial power and is considered one of the most important objects in the museum; however, the piece has been facing for decades a gradual deterioration, mainly in its fabrics. With the growing motivation to preserve this property, conservation and restoration processes were conducted in all parts of the throne. As a part of this treatment, a microbiological survey was conducted in selected parts of the object and the surrounding environment where it was placed. This biodeterioration study, control and detection of microbial species indicated a very pertinent answer in relation to the place it was exposed, and the restoration process was performed in order to solve the most pronounced damages aiming to restore the original characteristics of the artefact. The study indicated a high diversity of fungi and bacteria. It was observed a marked decrease, before and after restoration, being observed the recurrence of the genera Absidia, Cladosporium and Epicoccum. This reduction was not so pronounced for bacteria, being also observed the recurrence of some bacterial groups after restoration (Microbacterium, Staphylococcus and Kocuria). In the atmosphere new fungal species were found after restoration, indicating a natural recontamination of the exhibition area.
Downloads
References
[1] Zanatta, E. M., ‘Subjetividade e objetividade: as decisões nos processos de conservação e restauração dos bens culturais’, Tese de Doutoramento, Universidade do Federal do Estado do Rio de Janeiro/UNIRIO e Museu de Astronomia e Ciências Afins/MAST, Rio de Janeiro (2017).
[2] Pasquarella, C.; Balocco, C.; Pasquariello, G.; Petrone, G.; Saccani, E.; Manotti, P.; Ugolotti. M.; Palla, F.; Maggi, O.; Albertini, R., ‘A multidisciplinary approach to the study of cultural heritage environments: Experience at the Palatina Library in Parma’, Science of the Total Environment 536 (2015) 557-567, https://doi.org/10.1016/j.scitotenv.2015.07.105.
[3] Wenzel, M.; Schönig, I.; Berchtold, M.; Kämpfer, P.; König, H., ‘Aerobic and facultatively anaerobic cellulolytic bacteria from the gut of the termite Zootermopsis angusticollis’, Journal of Applied Microbiology 92(1) (2002) 32-40, https://doi.org/10.1046/j.1365-2672.2002.01502.x.
[4] Di Carlo, E.; Chisesi, R.; Barresi, G.; Barbaro, S.; Lombardo, G.; Rotolo, V.; Sebastianelli, M.; Travagliato, G.; Palla, F., ‘Fungi and bacteria in indoor cultural heritage environments: microbial-related risks for artworks and human health’, Environment and Ecology Research 4(5) (2016) 257-264, https://doi.org/10.13189/eer.2016.040504.
[5] Sterflinger, K.; Piñar, G., Microbial deterioration of cultural heritage and works of art – tilting at windmills?’, Applied Microbiology and Biotechnology 97 (2013) 9637-9646, https://doi.org/10.1007/s00253-013-5283-1.
[6] Akinyale, H. A., ‘Isolation of bacteria with potential for production of thermostable extracellular cellulase from sawdust’, Tese (Doutorado), Faculty of Science, University of Ibadan (2014).
[7] Gerber, E.; Bernard, R.; Castang, S.; Chabot, N.; Coze, F.; Dreux-Zigha, A.; Hauser, E.; Hivin, P.; Joseph, P.; Lazarelli, C.; Letellier, G.; Olive, J.; Leonetti, J. P., ‘Deinococcus as new chassis for industrial biotechnology: biology, physiology and tools’, Journal of Applied Microbiology 119(1) (2015) 1-10, https://doi. org/10.1111/jam.12808.
[8] Li, Q.; Zhang, B.; Yang, X.; Ge, Q., ‘Deterioration-associated microbiome of stone monuments: structure, variation, and assembly’, Applied Environmental Microbiology 84(7) (2018) 2680-2687, https://doi.org/10.1128/AEM.02680-17.
[9] Johnson, T. A.; Sims, G. K.; Ellsworth, T. R.; Ballance, A. R., ‘Effects of moisture and sorption on bioavailability of p-hydroxybenzoic acid to Arthrobacter sp. in soil’, Microbiological Research 153(4) (1999) 349-353, https://doi.org/10.1016/S0944-5013(99)80049-4.
[10] Mongodin, E. F.; Shapir, N.; Daugherty, S. C.; Deboy, R. T.; Emerson, J. B.; Shvartzbeyn, A.; Radune, D.; Vamathevan, J.; Riggs, F.; Grinberg, V.;Khouri, H.; Wackett, L. P.; Nelson, K. E.; Sadowsky, M. J., ‘Secrets of soil survival revealed by the genome sequence of Arthrobacter aurescens TC1’, PLoS Genetics 2(12) (2006) 2094-2106, https://doi.org/10.1371/journal.pgen.0020214.
[11] Marvasi, M.; Vedovato, E.; Balsamo, C.; Macherelli, A.; Dei, L.; Mastromei, G.; Perito, B., ‘Bacterial community analysis on the Mediaeval stained glass window “Natività” in the Florence Cathedral’, Journal of Cultural Heritage 10(1) (2009) 124-133, https://doi.org/10.1016/j.culher.2008.08.010.
[12] Suihko, M. L.; Alakomi, H. L.; Gorbushina, A.; Fortune, I.; Marquardt, J.; Saarela, M., ‘Characterization of aerobic bacterial and fungal microbiota on surfaces of historic Scottish monuments’, Systematic and Applied Microbiology 30(6) (2007) 494-508, https://doi.org/10.1016/j.syapm.2007.05.001.
[13] Gutarowska, B.; Skora, J.; Zduniak, K.; Rembisz, D., ‘Analysis of the sensitivity of microorganisms contaminating museums and archives to silver nanoparticles’, International Biodeterioration & Biodegradation 68 (2012) 7-17, https://doi.org/10.1016/j.ibiod.2011.12.002.
[14] Saarela, M.; Alakomi, H. L.; Suihko, M. L.; Maunuksela, L.; Raaska, L.; Mattila- Sandholm, T., ‘Heterotrophic microorganisms in air and biofilm samples from Roman catacombs, with special emphasis on actinobacteria and fungi’, International Biodeterioration & Biodegradation 54(1) (2004) 27-37, https://doi.org/10.1016/j.ibiod.2003.12.003.
[15] Wang, W.; Ma, Y.; Ma, X.; Wu, F.; Ma, X.; An, L.; Feng, H., ‘Diversity and seasonal dynamics of airborne bacteria in the Mogao Grottoes, Dunhuang, China’, Aerobiologia 28(1) (2012) 27-38, https://doi.org/10.1007/s10453-011-9208-0.
[16] Heyrman, J.; Swings, J., ‘16S rDNA Sequence analysis of bacterial isolates from biodeteriorated mural paintings in the Servilia Tomb (Necropolis of Carmona, Seville, Spain)’, Systematic and Applied Microbiology 24(3) (2001) 417-422, https://doi.org/10.1078/0723-2020-00048.
[17] Lutterbach, M. T. S.; Oliveira, A.L.C.; Zanatta, E.; da Costa, A. C. A., ‘A berlinda de aparato do imperador D. Pedro II: identificação de fungos em partes selecionadas e sua relação com biodeterioração e aerobiologia’, Conservar Património 17 (2013) 59- 72, https://doi.org/10.14568/cp2013003.
[18] da Costa, A. C. A.; Corrêa, F. N.; Lino, L. A. S.; Almeida, E. H. P.; Oliveira, A. L. C.; Lutterbach, M. T. S., 'Microbiological characterization of contaminating cells on scientific collections in a specialized library', Annual Research & Review in Biology 4 (2014) 3915-3931, https://doi.org/10.9734/ARRB/2014/11395.
[19] Kim, M. J.; Shin, H. K.; Choi, Y. S.; Kim, G. C.; Kim, G. H., ‘An aeromycological study of various wooden cultural heritages in Korea’, Journal of Cultural Heritage 17 (2016) 123-130, https://doi.org/10.1016/j.culher.2015.05.001.
[20] Coutinho, M. L.; Miller, A. Z.; Macedo, M. F., 'Biological colonization and biodeterioration of architectural ceramic materials: An overview', Journal of Cultural Heritage 16 (2015) 759-777, https://doi.org/10.1016/j.culher.2015.01.006.
[21] Ruga, L.; Orlandi, F.; Romano, B.; Fornaciari, M. ‘The assessment of fungal bioaerosols in the crypt of St. Peter in Perugia (Italy)’, International Biodeterioration & Biodegradation 98 (2015) 121-130, https://doi.org/10.1016/j.ibiod.2014.12.010.
[22] Do Campo, S.; Trigo, M. M.; Recio, M.; Melgar, M.; García-Sanchez, J.; Cabezudo, B., 'Fungal spore content of the atmosphere of the Cave of Nerja (southern Spain): diversity and origin', Science of the Total Environment 409 (2011) 835-843, https://doi.org/10.1016/j.scitotenv.2010.10.048.
[23] Porca, E.; Jurado, V.; Martin-Sanchez, P. M.; Hermosín, B.; Bastian, F.; Alabouvette, C.; Sáiz-Jiménez, C., ‘Aerobiology: an ecological indicator for early detection and control of fungal outbreaks in caves’, Ecological Indicators 11(6) (2011) 1594-1598, https://doi.org/10.1016/j.ecolind.2011.04.003.
[24] Martin-Sanchez, P. M.; Bastian, F.; Alabouvette, C.; Saiz- Jimenez, C., ‘Real-time PCR detection of Ochroconis lascauxensis involved in the formation of black stains in the Lascaux Cave, France’, Science of the Total Environment 443 (2013) 478-484, https://doi.org/10.1016/j.scitotenv.2012.11.026.
Downloads
Published
How to Cite
Issue
Section
Categories
License
This work is distributed under a Creative Commons Attribution License (CC BY-NC-ND 4.0) which permits use, distribution, and reproduction in any medium following no commercial or derivatives, provided the original author and source are credited.
Copyright remains with the authors.